Jacques Charles was born on the 12 of November 1746 in Beaugency, France. When he was young he had an education that consisted of very basic maths and hardly any practical science. Charles was fascinated with aeronautics when he first saw the Montgolfier brothers launch their balloon in early June 1783. Charles then persisted to make his own balloon with hydrogen gas produced from the reaction with iron filings and sulphuric acid. 10 years later with the help of his brothers, he successfully became the first person to fly in a hydrogen filled balloon to attain a maximum height of about 3000 meters (Jacques Charles, 2013).
Charles is best known for his law on gases stating how the volume of gases changes with temperature. This means that the warmer a gas gets the larger its volume, and the colder a gas gets the smaller its volume. Charles studied the gases oxygen, nitrogen, hydrogen and carbon dioxide at constant pressures and found that they all identically increased in volume as the temperature was raised. Charles however never published his findings but because Joseph Gay-Lussac had witnessed him conducting his experiments and re-constructed them for himself, he acknowledged Charles for the original idea (Blauch, Gas Laws: Charles' Law, 2012). Charles’ law (the temperature-volume law) states that “the volume of a given amount of gas held at constant pressure is directly proportional to the Kelvin temperature” (quote sourced from http://chemistry.bd.psu.edu/jircitano/gases.html )
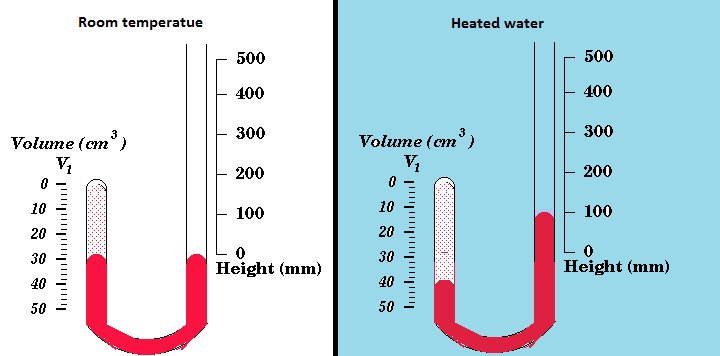
For his experiment, just like Boyle did, Charles used a J-shaped tube with a certain amount of gas trapped inside it. Mercury was then added and allowed to equalise in height at the bottom of the tube. With both levels of mercury at the same height ensured that the pressure inside the tube was the same as the atmospheric pressure. The tube was then submerged in water and by varying the temperature of the water, it changed the temperature of the gas within the tube. This showed that when the temperature was raised the total volume of the gas would rise, pushing the mercury up the j-shape tube. Using this experiment the relationship between volume and temperature of a gas can be seen (Charles' Law).
Charles is best known for his law on gases stating how the volume of gases changes with temperature. This means that the warmer a gas gets the larger its volume, and the colder a gas gets the smaller its volume. Charles studied the gases oxygen, nitrogen, hydrogen and carbon dioxide at constant pressures and found that they all identically increased in volume as the temperature was raised. Charles however never published his findings but because Joseph Gay-Lussac had witnessed him conducting his experiments and re-constructed them for himself, he acknowledged Charles for the original idea (Blauch, Gas Laws: Charles' Law, 2012). Charles’ law (the temperature-volume law) states that “the volume of a given amount of gas held at constant pressure is directly proportional to the Kelvin temperature” (quote sourced from http://chemistry.bd.psu.edu/jircitano/gases.html )
For his experiment, just like Boyle did, Charles used a J-shaped tube with a certain amount of gas trapped inside it. Mercury was then added and allowed to equalise in height at the bottom of the tube. With both levels of mercury at the same height ensured that the pressure inside the tube was the same as the atmospheric pressure. The tube was then submerged in water and by varying the temperature of the water, it changed the temperature of the gas within the tube. This showed that when the temperature was raised the total volume of the gas would rise, pushing the mercury up the j-shape tube. Using this experiment the relationship between volume and temperature of a gas can be seen (Charles' Law).
If a piston sitting 10cm high filled with 50ml of gas at 25 degrees celcius, how much warmer would the air inside need to be to raise the piston to 25cm. 25cm/10cm = 2.5, 50ml x 2.5 = 125ml, therefore 50ml / 298.15K = 125ml / T2, = 125ml x 298.15 / 50ml = 745.375 K = 472.225 degrees celcius. A piston like this, while unpractical due to heating and cooling times could be used to raise any number of objects as long as the equation was changed accordingly to suit the extra weight.